 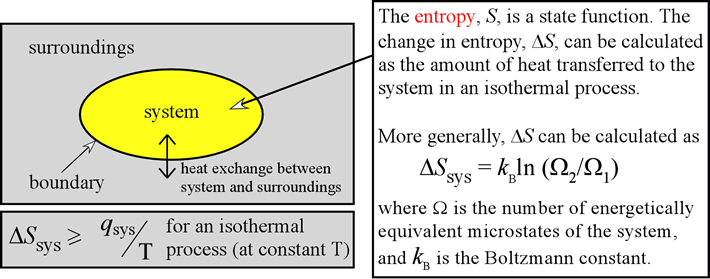 
For a given physical process, the entropy of the system and the environment will remain a constant if the process can be reversed. The change in entropy (delta S) is equal to the heat transfer (delta Q) divided by the temperature (T). If it’s negative and it will not happen, end of story.and it will not happen, end of story. The second law states that there exists a useful state variable called entropy. If it’s negative reaction will happen spontaneously. If the change in entropy of the universe is positive change in entropy of the universe is positive and the and the reaction will happen spontaneously. Entropy refers to the measure of the level of disorder in a thermodynamic system. A reaction is favoured if the enthalpy of the system decreases over the reaction. The enthalpy change of a reaction is equivalent to the amount of energy lost or gained during the reaction. If the plus the change in entropy of the surroundings. Key Facts & Summary: Enthalpy is the heat content of a system. ΔΔSSunivuniv = ΔΔSSsyssys + + ΔΔSSsurrsurrįor any reaction the change in entropy of the universe is For any reaction the change in entropy of the universe is going to be equal to the change in entropy of our system going to be equal to the change in entropy of our system plus the change in entropy of the surroundings. Solution: Entropy is to measure the randomness of activity in a system, on the other hand, enthalpy is the measurement of the overall amount of energy in the system. Shiva is happy = Shiva is happy = Shiva’s lawShiva’s law Universe is constant but the Entropy of the universe is constant but the Entropy of the Universe is always increasing.Universe is always increasing. This enthalpy change cannot be measured directly so a Born-Haber cycle is used to determine its value. Denoted as S, the change of entropy suggests that time itself is asymmetric with respect to order of an isolated system, meaning: a system will become more disordered, as time increases. Entropy (Shiva)We know that the amount of energy in the We know that the amount of energy in the The thermodynamic arrow of time (entropy) is the measurement of disorder within a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
